ELISA Technological Innovation: Core Methods, Innovative Progress and Global Applications
1
In-Depth Analysis of the Four Classic ELISA Types
The Enzyme-Linked Immunosorbent Assay (ELISA) is a fundamental immunochemical method celebrated for its high specificity, sensitivity, and relative ease of operation. Its core mechanism is based on the specific binding between antigens and antibodies, with detection accomplished through an enzyme attached to an antibody—this enzyme catalyzes a colorimetric or chemiluminescent reaction when exposed to a substrate, enabling both qualitative and quantitative analysis of target molecules. As a key tool, ELISA occupies a central position in various fields, such as basic scientific research, clinical diagnostics, environmental monitoring, and pharmaceutical development.
Although there are numerous ELISA variants, they are all essentially derived from four classic formats: Direct, Indirect, Sandwich, and Competitive ELISA. Gaining a thorough understanding of these types is vital for selecting the proper experimental protocol, correctly interpreting results, and maximizing the potential of this versatile technique.
1.1 Direct ELISA
Direct ELISA utilizes only an enzyme-labeled primary antibody. This enzyme-conjugated primary antibody binds "directly" to the antigen in the sample that has been immobilized on the plate. Subsequently, the enzyme-labeled primary antibody reacts with its substrate to produce a visible, measurable signal, whose intensity is directly related to the quantity of antigen present in the sample.
1.2 Indirect ELISA
Indirect ELISA makes use of both an unlabeled primary antibody and an enzyme-conjugated secondary antibody. The primary antibody first binds to the antigen immobilized on the plate, and the enzyme-conjugated secondary antibody then attaches to the primary antibody. Finally, the enzyme linked to the secondary antibody reacts with the substrate to generate a visible, detectable signal.
1.3 Sandwich ELISA
Unlike direct and indirect ELISA (where antigens are immobilized on the plate), sandwich ELISA employs two antibodies that bind to separate, non-overlapping epitopes on the target antigen. The "capture antibody" is pre-coated on the plate to immobilize the antigen from the sample. In indirect Sandwich ELISA, an unlabeled primary detection antibody is added and binds to the captured antigen, forming an antibody-antigen-antibody "sandwich". An enzyme-conjugated secondary antibody is then added, and a visible signal is produced when the substrate is introduced. The intensity of this signal is directly correlated with the amount of captured antigen.
1.4 Competitive ELISA
Competitive ELISA, also referred to as inhibition ELISA, involves the target antigen competing with a plate-immobilized reference antigen for binding to a limited amount of enzyme-labeled antibody. The quantity of labeled antibody that binds to the plate is inversely proportional to the concentration of antigen in the sample. Thus, a higher concentration of antigen in the sample leads to less antibody binding and a weaker measured signal after substrate addition.
There is another form of competitive ELISA based on antigen capture, in which the plate is coated with an unlabeled antibody. In this case, a known amount of enzyme-labeled antigen and the antigen in the sample compete for binding to the pre-coated antibody.
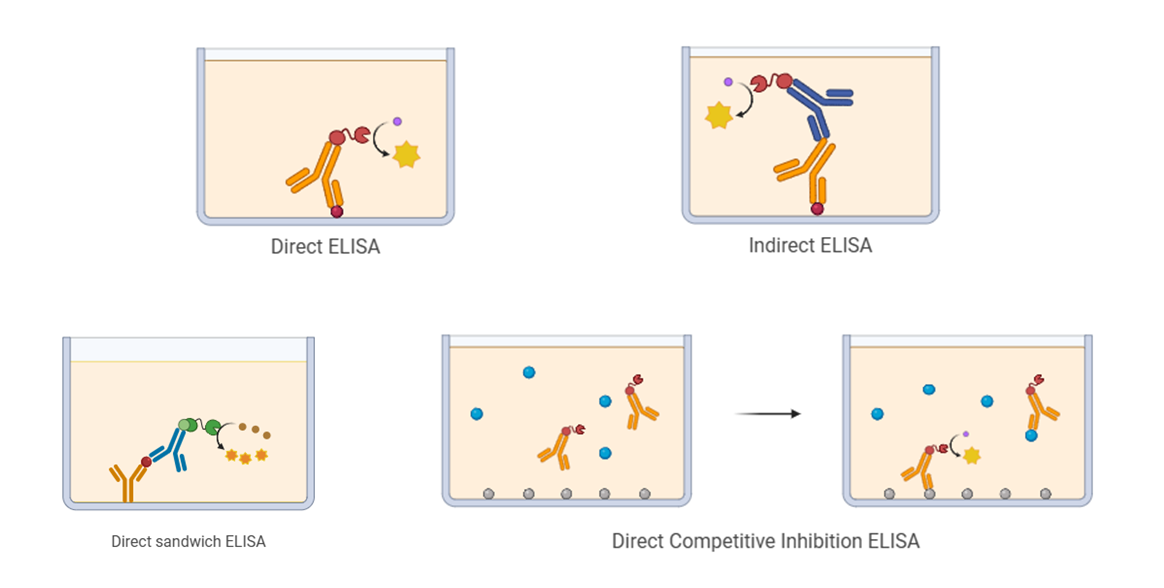
Table 1. Advantages and disadvantages of each ELISA type
| Type | Advantages | Disadvantages |
|---|---|---|
| Direct ELISA | Simple protocol, time-saving, reagents-saving; No cross-reactivity from secondary antibody | High background; No signal amplification; Low flexibility (requires conjugated primary antibody) |
| Indirect ELISA | Signal amplification; Higher sensitivity; High flexibility (shared secondary antibody) | More complex protocol; Cross-reactivity risk |
| Sandwich ELISA | High flexibility; High sensitivity; High specificity | Requires large antigen with ≥2 epitopes; Difficult to find matched antibody pairs |
| Competitive ELISA | High flexibility; High sensitivity; Ideal for small antigens/haptens | Relatively complex; Requires inhibitor antigen |
Selection Decision Guide
The choice of the optimal ELISA format depends on the nature of the analyte, sensitivity requirements, available antibodies, throughput, and cost.
- Sandwich ELISA: The first choice for detecting specific proteins in complex samples
- Indirect ELISA: Versatile for antibody detection or antigen detection when only one antibody is available
- Direct ELISA: Suitable for high-throughput screening of purified antigens
- Competitive ELISA: The only option for detecting small molecules (signal is inversely correlated with concentration)
For detailed selection methods, please refer to our article How to Select the Appropriate ELISA Kit.
2
Innovations and Frontiers in ELISA Technology
Recent advancements in ELISA technology since 2020 have focused on redesigning assay structures, incorporating powerful biochemical amplification components, and moving from simple colorimetric plate readers to multiplexed optical, electrochemical, and AI-assisted detection systems.
2.1 ELISA design: architectures, microfluidics, and digital formats
Research focus has shifted from static 96-well plates to integrated microfluidic and digital-microfluidic (DMF) formats, which automate experimental workflows, reduce sample volume, and support high-throughput multiplexing. Electrowetting-on-dielectric (EWOD) chips perform all key ELISA steps on droplets, enhancing sensitivity and decreasing reagent usage. Integrating DMF with droplet ELISA allows for single-molecule detection by compartmentalizing individual immunocomplexes into tiny droplets.
2.2 Signal amplification: synthetic biology, nanomaterials, and in situ generation
Modern ELISA development increasingly treats immunocomplexes as triggers for programmable biochemical amplification, rather than direct links to a single enzyme reaction. Expression immunoassays, CRISPR-linked immunoassays (CLISA), and T7 RNA polymerase–linked immunosensing assays connect antigen recognition to transcription or translation cascades, pushing detection limits to the low-femtomolar or attomolar range.
2.3 Detection systems: from colorimetric to multiplexed, digital, and AI-assisted readouts
Detection methods have evolved from simple absorbance measurements to a variety of optical, electrochemical, and digital readouts, designed to enhance sensitivity, enable multiplexing, and increase automation.
2.4 Other: Paper-based, portable, and point-of-care ELISA
Paper-based ELISA (p-ELISA) with multilayer "merry-go-round" designs enables automatic, electricity-free assays suitable for decentralized testing. Portable point-of-care (POC) devices combine microfluidics, dried reagents, and smartphone readers for field deployment, making them ideal for diagnosing infectious and chronic diseases in resource-limited settings.
3
Evolving Applications of ELISA: From Lab Bench to Real World
ELISA has expanded well beyond basic research laboratories to become a cornerstone technology in clinical diagnostics, public health, food safety, environmental monitoring, biothreat detection, and point-of-care testing worldwide.
3.1 Clinical diagnostics and precision medicine
ELISA is widely employed for screening, diagnosis, prognosis, and therapeutic monitoring:
- Infectious diseases: Serves as the gold standard for serological testing of HIV, HBV/HCV, syphilis, Lyme disease, H. pylori, influenza, and SARS-CoV-2
- Cancer biomarkers: Quantifies key circulating tumor markers such as PSA, CEA, CA125, CA19-9, and AFP
- Hormones: Reliably measures thyroid hormones, reproductive hormones, growth factors, and stress hormones
- Autoimmunity: Detects autoantibodies associated with rheumatoid arthritis, systemic lupus erythematosus, multiple sclerosis, and celiac disease
- Therapeutic drug monitoring (TDM): Quantifies levels of monoclonal antibodies, antibiotics, and immunosuppressants
3.2 Public health and epidemic surveillance
ELISA plays an indispensable role in outbreak response, seroprevalence surveys, vaccination monitoring, and disease control programs:
- Seroprevalence: Provides population-level data on past infection and immunity
- Vaccination: Measures antibody titers post-vaccination to assess vaccine immunogenicity
- Neglected tropical diseases: Widely used for screening and surveillance in low- and middle-income countries
- Integrated networks: National and global health agencies use ELISA-based data to track disease trends
3.3 High-plex and high-throughput ELISA-like platforms
Next-generation ELISA architectures focus on high plex and throughput while maintaining the specificity of traditional ELISA. Bead-based nELISA enables high-throughput, high-plex protein quantification, supporting biomarker discovery, immune monitoring, and clinical diagnostics.
3.4 Food safety and environmental monitoring
ELISA is a standard analytical tool for detecting contaminants, allergens, pathogens, residues, and GMOs across various sectors:
- Pathogens: Screens for Salmonella, Listeria, E. coli O157:H7, and Campylobacter
- Allergens: Identifies trace levels of peanut, milk, egg, wheat, soy, fish, and shellfish allergens
- Mycotoxins: Quantifies aflatoxins, ochratoxin A, and fumonisins in grains, nuts, and dairy products
- Veterinary residues: Monitors antibiotics, hormones, and other veterinary pharmaceuticals
- Water/soil: Detects microbes, toxins, pesticides, heavy metals, and endocrine disruptors
- Biothreats: Enables rapid field identification of high-consequence biothreat agents
3.5 Integration with AI and predictive diagnostics
ELISA is increasingly integrated into AI-assisted diagnostic frameworks. Traditional ELISAs typically require 3–5 hours to complete; new platforms combine immunoassay measurements with deep learning models to support early clinical decision-making, enhancing the utility of classical ELISA with advanced computational tools.
4
Conclusion and Future Perspectives
The four fundamental ELISA types—direct, indirect, sandwich, and competitive—each offer unique and irreplaceable value, forming a versatile platform that has driven decades of progress in scientific and clinical research. The continuous innovation of ELISA, through integration with microfluidics, digital readouts, nanotechnology, and AI, demonstrates its remarkable adaptability. In the future, ELISA will become faster, more sensitive, intelligent, and accessible, playing an increasingly critical role in precision medicine, global public health, and environmental stewardship. ExKits remains committed to advancing ELISA technology and expanding its applications to address global health and environmental challenges.
