How to Solve Common Troubles in ELISA Experiments
Enzyme-linked immunosorbent assay, also called ELISA or EIA, is a plate-based testing method used to detect and quantify peptides, proteins, antibodies, hormones and other substances. It identifies target substances through specific antibody binding and color development reactions. In an ELISA test, antigens are immobilized on a solid-phase surface and then combined with enzyme-labeled antibodies. Detection is achieved by incubating with a substrate to measure the product generated by enzyme catalytic activity, and the core of the detection system lies in highly specific antigen-antibody binding. ELISA is a commonly used blood antibody detection assay, which can be applied to assist in the diagnosis of various diseases, including HIV (the pathogen causing AIDS), Lyme disease, pernicious anemia, Rocky Mountain spotted fever, rotavirus, squamous cell carcinoma, syphilis and others.
ExKits owns a complete ELISA kit development platform and a mature antigen-antibody R&D system. The team is proficient in diverse ELISA technologies, including double-antibody sandwich assay, double-antigen sandwich assay, direct competitive ELISA, indirect competitive ELISA, blocking assay, indirect ELISA and other methods. Cooperating with a professional diagnostic kit development team, ExKits can develop kits that meet clinical diagnostic standards, with product quality reaching an advanced level.
This article mainly summarizes the common problems encountered in ELISA experiments and provides corresponding solutions.
1
What is the common procedure of ELISA?
ELISA is short for enzyme-linked immunosorbent assay, which is used to confirm the existence of specific proteins and determine their concentration levels. There are four main types of ELISA: sandwich ELISA, competitive ELISA, indirect ELISA and direct ELISA. The standard experimental steps include reagent storage, reagent preparation, plate preparation, sample/reagent addition, incubation, washing and reading. As shown in Figure 1, the detailed competitive ELISA experimental steps are presented.
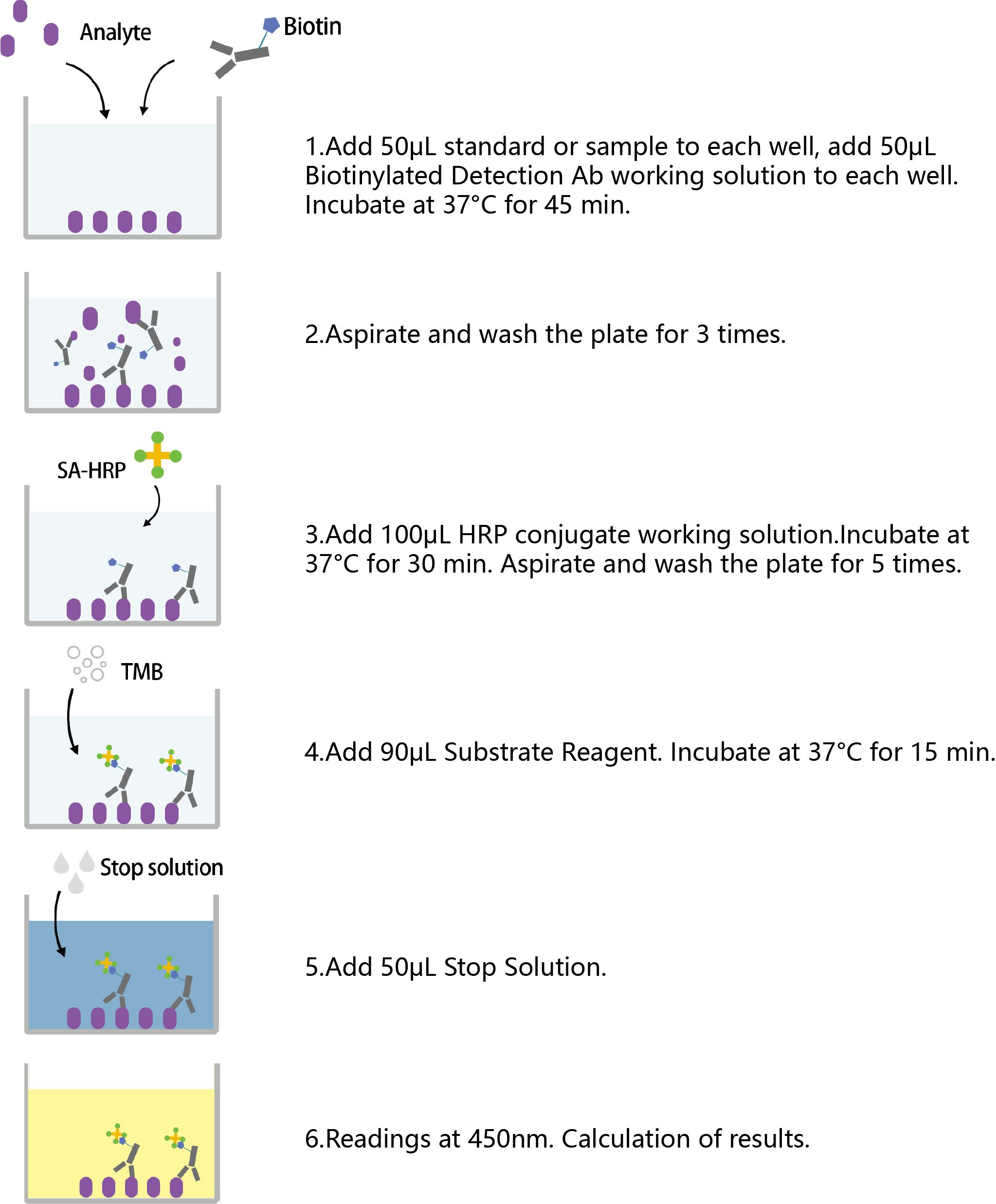
Figure 1. The procedures of competition ELISA
2
Several problems of the color reaction in ELISA experiment
ELISA results are usually judged by the color depth of the chromogenic substrate reaction. Generally, the color reaction is carried out at 37 °C for about 15 minutes, then terminated with stop solution, and the absorbance value is measured with a microplate reader at a specific wavelength. Since the detection optical path of each well is perpendicular to the microplate reader, the bottom of the reaction plate must be kept clean during detection, and the addition volume of chromogenic substrate and stop solution must be accurate, as the liquid volume in the well will directly affect the final reading. This section summarizes several common abnormal color reaction phenomena in ELISA experiments, and lists the causes and solutions in Table 1.
Table 1. The problems of the color reaction
No signal
| cause | solution |
|---|---|
| Incorrect experimental operation, wrong reagents or mismatched detection wavelength | Check the protocol carefully, repeat the experiment with positive control, and verify the wavelength, filter and gain settings of the microplate reader. |
| Insufficient antibody dosage | Adjust the concentration of primary antibody and/or secondary antibody for testing. |
| Over-short incubation time | Extend incubation to overnight at 4°C or follow the kit instructions strictly. |
| Antibody stored at 4°C for a long time or repeated freeze-thaw cycles | Use a new aliquot of antibody stored at -20°C or lower. |
| Epitope recognition blocked by plate adsorption | For peptide detection in direct or indirect ELISA, couple the peptide to a large carrier protein before coating the microplate. |
| Slow enzymatic color reaction | Prepare substrate solution fresh before use, confirm the validity and no contamination of stock solution, and extend incubation time appropriately. |
Low sensitivity or weak signal
| cause | solution |
|---|---|
| Improper storage of ELISA kit | Store all reagents in accordance with the recommended conditions; note that different reagents may have different storage requirements. |
| Wrong microplate reader parameter settings | Check and correct the wavelength, filter, gain and other parameters of the microplate reader. |
| Inactivated or over-diluted detection reagent | Confirm the activity of reporter enzyme/fluorescent substance, or increase the concentration of detection reagent. |
| Insufficient antigen coating on the microplate | Increase the dosage of coating antigen or optimize the coating buffer. |
| Insufficient antibody usage | Raise the concentration of primary antibody and/or secondary antibody, and optimize the antibody dilution ratio. |
| Mixing reagents from different kits | Do not mix components of different batches or types of kits. |
| Over-low incubation temperature | Optimize the incubation temperature; balance all reagents to room temperature before the experiment. |
| Damaged or reused ELISA plates | Store sealed plates in a desiccant-containing bag at low temperature to avoid moisture; equilibrate sealed plates to room temperature before use to prevent condensation. Mark used wells to avoid repeated use, seal them and use the remaining wells as soon as possible, and keep desiccant in the storage bag. |
High background signal
| cause | solution |
|---|---|
| Excessive antibody dosage | Optimize antibody concentration by gradient dilution test. |
| Excessive detection reagent | Ensure correct dilution of detection reagent, or increase the dilution ratio for retesting. |
| Over-high incubation temperature | Adjust and optimize the incubation temperature. |
| Reaction not terminated in time | Add stop solution to stop excessive color development. |
| Delayed reading after adding stop solution | Read the plate immediately after terminating the reaction. |
| Substrate incubation exposed to light | Keep away from light during substrate incubation. |
| Non-specific antibody binding | Use a suitable blocking buffer or affinity-purified antibody. |
| Contaminated plate bottom | Wipe the bottom of the plate clean and read again. |
Slow color development
| cause | solution |
|---|---|
| Incorrect incubation temperature | Keep plates and reagents at room temperature. |
| Contaminated solution | Prepare fresh working solution. |
| Expired, contaminated or pH-mismatched detection reagent | Use fresh detection reagent with correct pH value. |
| Wrong conjugate, improper preparation or degraded conjugate | Use the matching conjugate provided with the kit; each conjugate is specific to the kit and batch. If working conjugate needs preparation, mix concentrate and diluent in accurate volume, do not prepare in advance or store leftover solution. If ready-to-use, take only the required volume and do not return leftover to the stock bottle. |
| Sodium azide contained in wash buffer | Do not add sodium azide to the wash buffer. |
3
Other problems of ELISA experiment
In addition to abnormal color reactions, ELISA experiments also have common problems related to standard curve, data reading and experimental repeatability. This section lists several typical problems and solutions as follows.
Why does the standard curve look poorly?
The standard curve is the key basis for sample quantification in ELISA data analysis, so the preparation quality of standard products and curves is crucial. The common causes and solutions for poor standard curve are as follows:
a. Improper standard dilution: Check and prepare standard gradient dilution accurately.
b. Incorrect standard reconstitution: Centrifuge the standard vial slightly before opening, and check for undissolved substances after reconstitution.
c. Degraded standard: Store and operate standard products in accordance with instructions.
d. Unsuitable curve fitting scale: Try different fitting methods such as log-log coordinate or five-parameter logistic curve.
e. Pipetting error: Use calibrated pipettes and standard pipetting techniques.
a. Improper standard dilution: Check and prepare standard gradient dilution accurately.
b. Incorrect standard reconstitution: Centrifuge the standard vial slightly before opening, and check for undissolved substances after reconstitution.
c. Degraded standard: Store and operate standard products in accordance with instructions.
d. Unsuitable curve fitting scale: Try different fitting methods such as log-log coordinate or five-parameter logistic curve.
e. Pipetting error: Use calibrated pipettes and standard pipetting techniques.
Why does it read low with normal results by eyes?
Wrong filter selection during reading. For TMB substrate, the detection wavelength should be 450nm with 650nm reference correction. Recheck the wavelength, filter and gain settings of the microplate reader before reading.
Why is repeatability poor?
a. Improper kit storage: Store all components according to the manual, avoid long-term placement at room temperature.
b. Non-standard preparation of standard: Reconstitute standard with the specified diluent strictly following the protocol; prepare reagents within 10 minutes before use and add them quickly.
c. Insufficient mixing after sample addition: Mix well with a vortex mixer when adding multiple reagents, and avoid splashing.
d. Uncalibrated microplate reader: Calibrate the microplate reader regularly.
e. Inconsistent conditions: Keep incubation time, washing, color development and operators consistent; retest the standard curve.
f. Inadequate washing: Add 200μL wash buffer to each well or fill the wells without overflow; check the washer nozzle for blockage to ensure sufficient washing.
g. Unstable temperature: Maintain constant temperature during incubation to avoid fluctuation.
h. Residue on well wall or well bottom scratched: Add sample slowly along the well wall, do not touch the well bottom with pipette tip.
i. Reused consumables: Replace pipette tips between samples and reagent reservoirs.
j. Values near the cutoff: Set three duplicate wells for each sample, and take consistent results of more than two wells.
k. Cross-contamination during sample addition: Avoid cross-contamination when adding samples.
b. Non-standard preparation of standard: Reconstitute standard with the specified diluent strictly following the protocol; prepare reagents within 10 minutes before use and add them quickly.
c. Insufficient mixing after sample addition: Mix well with a vortex mixer when adding multiple reagents, and avoid splashing.
d. Uncalibrated microplate reader: Calibrate the microplate reader regularly.
e. Inconsistent conditions: Keep incubation time, washing, color development and operators consistent; retest the standard curve.
f. Inadequate washing: Add 200μL wash buffer to each well or fill the wells without overflow; check the washer nozzle for blockage to ensure sufficient washing.
g. Unstable temperature: Maintain constant temperature during incubation to avoid fluctuation.
h. Residue on well wall or well bottom scratched: Add sample slowly along the well wall, do not touch the well bottom with pipette tip.
i. Reused consumables: Replace pipette tips between samples and reagent reservoirs.
j. Values near the cutoff: Set three duplicate wells for each sample, and take consistent results of more than two wells.
k. Cross-contamination during sample addition: Avoid cross-contamination when adding samples.
Why is abnormal color developed?
a. Cross-contamination in manual washing: Remove liquid in time after filling wash buffer three times, and set proper soaking time for subsequent washing to reduce cross-contamination.
b. Cross-contamination when patting the plate: Use clean paper towels, avoid introducing foreign matters, and do not pat the same position repeatedly.
c. Sample contamination due to long storage: Keep samples fresh or store at low temperature.
d. Insufficient washing or residual liquid caused by washer blockage: Fill each well fully without overflow, check and unblock the washer nozzle.
e. Precipitation or cell interference from incomplete sample centrifugation: Centrifuge serum and plasma samples thoroughly.
f. Wrong wash buffer preparation or misuse of concentrated solution: Prepare wash buffer strictly according to the manual.
b. Cross-contamination when patting the plate: Use clean paper towels, avoid introducing foreign matters, and do not pat the same position repeatedly.
c. Sample contamination due to long storage: Keep samples fresh or store at low temperature.
d. Insufficient washing or residual liquid caused by washer blockage: Fill each well fully without overflow, check and unblock the washer nozzle.
e. Precipitation or cell interference from incomplete sample centrifugation: Centrifuge serum and plasma samples thoroughly.
f. Wrong wash buffer preparation or misuse of concentrated solution: Prepare wash buffer strictly according to the manual.
