ELISA: The Key to Opening a New Era of Biomedical Detection
ELISA: A Powerful Assistant in Modern Biomedicine
In the field of modern biomedicine, ELISA (Enzyme-Linked Immunosorbent Assay) technology is undoubtedly a pivotal detection method, widely applied in disease diagnosis, drug development, food safety testing, environmental monitoring, and many other areas. It is like a precise "detective" that can sensitively and specifically detect various biomolecules, providing crucial information for researchers and clinicians. Today, let us embark on a journey through the development history of ELISA technology to explore how it has gradually become a powerful assistant in the biomedical field.
The development history of ELISA technology is a story of scientific progress filled with innovation and breakthroughs. From its initial theoretical concept to its widespread application today, it has gradually matured through continuous exploration and improvement, bringing revolutionary changes to biomedical research and clinical diagnosis.
Exploration Stemming from RIA
The origins of ELISA can be traced back to the 1960s, when Radioimmunoassay (RIA) technology was an important means for detecting trace substances in the biomedical field. In 1960, Rosalyn S. Yalow and Solomon A. Berson first used iodine-labeled RIA technology to successfully determine endogenous plasma insulin levels. This pioneering work opened a new door for the quantitative detection of biomolecules, earning Yalow the Nobel Prize in Physiology or Medicine in 1977.
However, RIA technology had obvious limitations. Using radioactive isotopes as markers required special protective equipment and strict operating protocols to ensure safety, and the disposal of radioactive waste was extremely, limiting its widespread application in more scenarios. With the growing demands of scientific research, scientists urgently needed to find a safer, more convenient alternative.
Breakthrough in Enzyme-Linked Immunoassays
In 1966, scientists achieved a crucial breakthrough. Researchers such as Perlmann and Schuurs boldly attempted to use enzymes instead of radioactive isotopes, conducting enzyme-linked immunoassays. They successfully demonstrated the feasibility of this method, laying a solid foundation for the birth of ELISA technology. This innovative idea was like a ray of light, illuminating a new direction for biological detection technology. Although many were skeptical at the time about whether enzymes could stably link to antibodies or antigens without affecting their biological activity, the research results of Perlmann and Schuurs strongly responded to these doubts.
Birth: A Moment That Changed the Detection Landscape
1971: ELISA Officially Emerges
After years of research and exploration, 1971 became a milestone year in the development history of ELISA technology. In this year, two groups of scientists led by Perlmann and Schuurs almost simultaneously published literature on ELISA, systematically introducing this new detection method to the world. Their papers detailed the principles, operating procedures, and application potential of ELISA in biomedical detection, laying a solid theoretical foundation for the widespread application of this technology.
The publication of this achievement was like a stone thrown into a calm lake, creating ripples in the biomedical field. With its unique advantages, ELISA technology quickly attracted the attention of numerous researchers and clinicians. It not only overcame the radioactive safety hazards of RIA technology but also boasted many advantages such as simple operation, high sensitivity, and strong specificity, bringing new hope to biomolecule detection.
Early Applications and Promotion
After its birth, ELISA quickly found applications in basic medical theoretical research, clinical virology, and biochemical testing due to its significant advantages. In basic medical theoretical research, researchers used ELISA technology to deeply explore the pathogenesis of various diseases, providing a more in-depth theoretical basis for disease prevention and treatment. For example, by detecting specific cytokine or antibody levels, researchers can better understand the role of the immune system in the occurrence and development of diseases.
In the field of clinical virology, ELISA became an important means for detecting viral infections. Taking hepatitis B virus detection as an example, ELISA technology can quickly and accurately detect hepatitis B virus markers in the blood, helping doctors timely diagnose whether patients are infected with hepatitis B virus and assess the severity of their condition. This provided strong support for the prevention and control of hepatitis B, greatly improving the efficiency and accuracy of disease diagnosis.
In biochemical testing, ELISA technology also played an important role. It can detect various biomolecules such as proteins, hormones, enzymes, etc., providing rich information for clinical diagnosis and treatment. For example, by detecting specific hormone levels in the blood, doctors can determine whether the patient's endocrine system is normal, thereby timely discovering and treating endocrine diseases.
Development: Technological Innovation Drives Progress
Expansion of Markers and Substrates
After the advent of ELISA technology, scientists did not stop their exploration but continued to improve and perfect it. In the early days of ELISA technology, the types of markers and substrates were relatively limited, which to some extent restricted the sensitivity and application range of the technology. To break through this limitation, researchers dedicated themselves to developing new markers and substrates.
In the late 1970s, various new markers such as alkaline phosphatase, horseradish peroxidase, etc., emerged one after another. These markers had more excellent performance than early markers, greatly improving the sensitivity and stability of ELISA detection. Take horseradish peroxidase as an example: it has high catalytic activity, good stability, and is easy to prepare and label, enabling it to more efficiently catalyze substrate reactions and produce obvious color changes, making detection results more accurate and reliable. Alkaline phosphatase also brought new vitality to ELISA technology with its unique catalytic properties.

In 1979, Wallac Company introduced an enzyme-linked immunosorbent assay method using fluorescein as a substrate, which laid a solid foundation for the development of quantitative ELISA. As a substrate, fluorescein emits fluorescence after being catalyzed by enzymes. By detecting the intensity of fluorescence, not only can the presence of target substances be qualitatively determined, but they can also be accurately quantified. The emergence of this method made the application of ELISA technology more extensive and in-depth in biomedical research, enabling researchers to more precisely measure the content of various substances in biological samples, providing more powerful data support for disease diagnosis, drug development, etc.
Emergence of Multiple ELISA Methods
With the continuous development of ELISA technology, various ELISA methods have emerged like bamboo shoots after a spring rain to meet different detection needs.
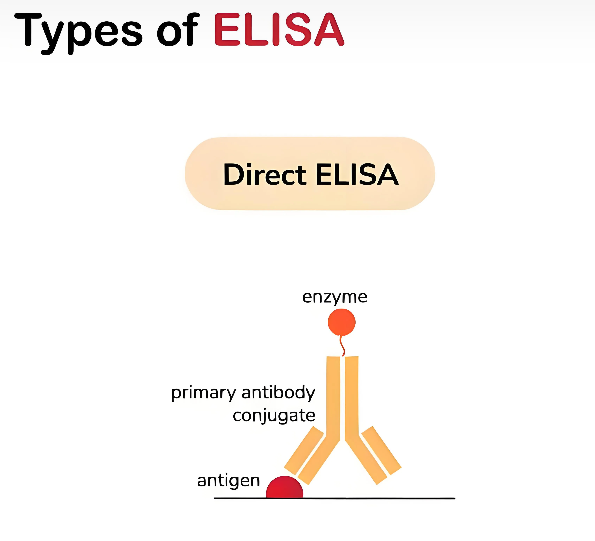
In 1971, Perlmann first established the Direct ELISA method. This method is usually used to detect relatively large antibodies or antigens, as well as for antibody screening and antigen epitope mapping. Its operation mode is relatively simple: the antigen is directly coated in the wells of a microplate, then enzyme-linked specific antibodies are added, which target the immobilized antigen to form a stable complex. Next, unbound antibodies or background proteins are washed away, then an appropriate amount of substrate is added and incubated to catalyze a specific enzyme reaction, producing a color reaction. The analyte can be quantified by detecting the fluorescence signal. The Direct ELISA method is like a direct "face-to-face" detection, simple and straightforward, but it requires primary antibodies to be pre-labeled, which limits its application range to a certain extent.
In 1978, inspired by the Direct ELISA method, Lindström and Wager published the Indirect ELISA method. Unlike the Direct ELISA method, the Indirect ELISA method introduces a second antibody for detection. In the experiment, the antigen is first immobilized on a solid phase carrier, then the antibody to be tested in the sample is added. After the primary antibody binds to the antigen, an enzyme-labeled secondary antibody is added, which can recognize and bind to the primary antibody, thereby amplifying the detection signal. Figuratively speaking, the Direct ELISA method is like a single light bulb illuminating the target, while the Indirect ELISA method is like connecting two light bulbs, greatly improving the sensitivity of detection. This method is mainly used to detect antibodies in samples, and since the enzyme-labeled antibody is universal, it can detect multiple different antibodies by simply changing the coating antigen, offering high flexibility.
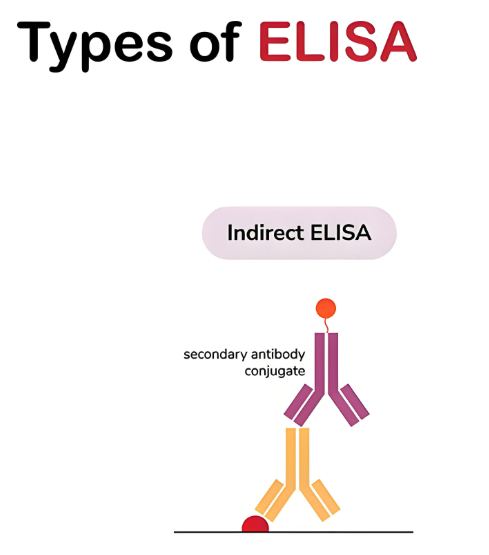
In 1977, Kato and his colleagues invented the Sandwich ELISA method, a classic method that combines the characteristics of Direct ELISA and Indirect ELISA technologies, and is also one of the most widely used ELISA methods today. "Sandwich" vividly describes its detection process: the method immobilizes a primary antibody on the well surface, then the test sample containing the target antigen is added to the well to fully bind to the antibody. After washing away unbound impurities, an enzyme-labeled secondary antibody is added, which binds to another epitope of the antigen, forming a structure like a "sandwich". The advantages of this method lie in its high specificity and sensitivity, just like using two slices of bread to precisely hold the cream in the middle, enabling accurate capture and detection of target antigens. It is suitable for detecting various macromolecular antigens, and is commonly used in medical testing to determine disease-related antigens such as HBsAg, HBeAg, AFP, and in scientific research to detect cytokines, etc.

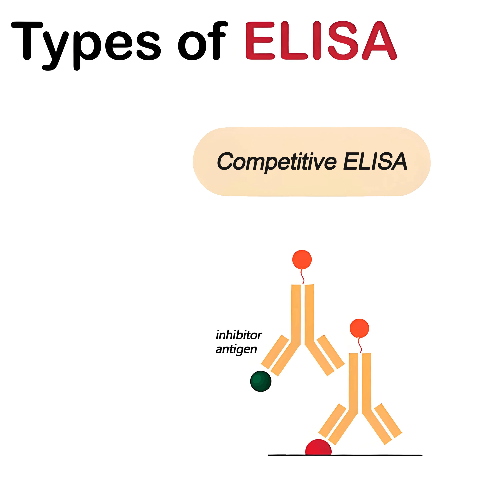
Competitive ELISA method is the most complex of all ELISA methods, mainly used to detect the intensity of interference signals to reflect the antigen level in the sample. Usually, enzyme-labeled antigen is used as the competitive antigen, which forms a direct competition with the antigen analyte from the sample to compete for binding sites on excess immobilized antibodies. Therefore, the higher the interference signal, the less antigen is present in the sample, which is similar to the "occupying seats" concept in daily life. When there is less antigen in the sample, the enzyme-labeled antigen can more easily bind to the immobilized antibody, thereby producing a stronger signal; conversely, when there is more antigen in the sample, they will occupy the binding sites of the immobilized antibody, reducing the binding opportunities of the enzyme-labeled antigen and correspondingly weakening the signal. This method is suitable for the detection of small molecule antigens and in cases where the sample contains interfering substances.
Impact: Reshaping the Biomedical Detection Landscape
Becoming the Industry "Gold Standard"
With its high sensitivity and high throughput, ELISA technology has stood out in many fields and become the "gold standard" in the industry. In the quantitative analysis of therapeutic antibody drugs, ELISA can accurately determine the concentration of antibody drugs, providing key data support for drug research and development, production, and clinical applications, ensuring the stability and reliability of drug quality and efficacy. For example, in the research and development process of tumor immunotherapy drugs, precise monitoring of therapeutic antibody concentrations through ELISA technology helps researchers evaluate the efficacy and safety of drugs, thereby promoting the development of new drugs.
In the food industry, ELISA technology is widely used to detect harmful substances in food, such as pesticide residues, veterinary drug residues, microbial toxins, etc. It can quickly and accurately detect trace pollutants in food, ensuring food safety and safeguarding consumer health. Taking the detection of melamine in milk as an example, ELISA technology can test a large number of milk samples in a short time, timely detect products with excessive melamine, and effectively prevent problem milk from entering the market.
In toxicology research, ELISA technology can detect toxic substances and their metabolites in biological samples, providing important basis for evaluating the toxicity and safety of chemical substances. Researchers analyze the levels of specific biomarkers in experimental animals through ELISA technology to understand the impact of chemical substances on organisms, providing scientific support for formulating reasonable safety standards and protective measures.
Promoting the Development of Related Fields
The emergence and development of ELISA technology have had a profound promoting effect on biomedical research and development, disease diagnosis, environmental monitoring and other fields.
In the field of biomedical research and development, ELISA technology provides a powerful tool for drug target screening, drug efficacy evaluation, and drug safety monitoring. By detecting changes in biomarkers before and after drug action, researchers can deeply understand the mechanism of drug action and accelerate the research and development process of new drugs. For example, in the research and development of diabetes drugs, using ELISA technology to detect the levels of insulin, blood glucose regulatory factors and other biomarkers in the blood helps evaluate the drug's effect on blood glucose regulation, thereby screening out effective drug candidates.
In disease diagnosis, ELISA technology has greatly improved the efficiency and accuracy of disease diagnosis. It can detect various disease-related biomarkers, such as tumor markers, viral antibodies, autoantibodies, etc., providing important basis for early disease diagnosis, condition monitoring and prognosis evaluation. Take AIDS diagnosis as an example: ELISA technology can detect HIV antibodies in the blood, helping doctors timely discover infected individuals and valuable time for patient treatment and management.
In the field of environmental monitoring, ELISA technology can be used to detect pollutants in soil, water bodies, and air, such as heavy metals, organic pollutants, microorganisms, etc. Through rapid and sensitive detection of environmental samples, environmental pollution problems can be timely discovered, providing scientific basis for environmental protection and governance. For example, using ELISA technology to detect the content of microcystin in water bodies can evaluate the eutrophication level of water bodies and provide decision support for water resource protection and management.

The development history of ELISA technology is a legend of technological innovation. From its initial theoretical concept to its widespread application in various fields today, it has brought revolutionary changes to biomedical research and clinical diagnosis, becoming an indispensable important tool in the modern biomedical field. It is believed that in the future, with the continuous advancement of science and technology, ELISA technology will continue to play an important role, making greater contributions to human health and environmental protection.
Review and Outlook:
Looking back at the development history of ELISA technology, from its birth to solve the drawbacks of RIA technology to its irreplaceable role in many fields today, every step of its progress has condensed the wisdom and efforts of countless researchers. The emergence of ELISA technology not only changed the way of biomedical detection but also promoted the development of the entire life science field. It has revealed the mysteries of the microcosm within organisms, allowing us to more deeply understand the pathogenesis of diseases, the effects of drugs, and the impact of the environment on organisms.
Looking forward to the future, with the rapid development of science and technology, ELISA technology is expected to make greater breakthroughs in the field of precision medicine. Through integration with emerging technologies such as gene sequencing and big data analysis, ELISA can achieve more precise disease diagnosis and personalized treatment plan formulation. For example, in cancer diagnosis, ELISA technology can more precisely detect early cancer markers, providing a valuable time window for patients' early treatment. At the same time, with the continuous deepening of biomarker research, ELISA technology will be able to detect more types of biomarkers, providing more comprehensive information for disease prediction, diagnosis, and treatment. It is believed that in the future, ELISA technology will continue to contribute more to human health and scientific research, becoming an important engine driving biomedical progress.
References: 1. Yalow RS, Berson SA. Immunoassay of endogenous plasma insulin in man. J Clin Invest. 1960;39(7). 2. Perlmann P, Holmgren E. Enzyme - linked immunosorbent assay (ELISA). III. Detection of antibodies in cancer patients. J Immunol Methods. 1971;4(2). 3. Schuurs AH, van Weemen BK. Immunoassay using antigen - enzyme conjugates. FEBS Lett. 1971;15(3). 4. Engvall E, Perlmann P. Enzyme - linked immunosorbent assay (ELISA). Quantitative assay of immunoglobulin G. Immunochemistry. 1972;9(10). 5. Voller A, Bidwell DE, Bartlett A. Enzyme - linked immunosorbent assay for the detection of antibodies. J Clin Pathol. 1976;29(12). 6. Lindström K, Wager O. An indirect enzyme - linked immunosorbent assay (ELISA) for the detection of antibodies against group A streptococci. J Immunol Methods. 1978;21(3). 7. Kato K, et al. Sandwich enzyme - linked immunosorbent assay for the detection of hepatitis B surface antigen. J Clin Microbiol. 1977;6(5). 8. Guilbault GG, Montalvo - Ramirez G. Fluorescence - based enzyme - linked immunosorbent assay. Anal Chem. 1979;51(11). |
