What Really Affects Your ELISA Results? A Complete Guide
In the complex field of biomolecular detection, the enzyme‑linked immunosorbent assay (ELISA) stands out as one of the most widely applied and influential techniques. As a core analytical method, it relies on the specific binding of antigens and antibodies, with signal amplification by enzymes, enabling highly sensitive and specific detection of targets including disease biomarkers and environmental pollutants. By converting molecular interactions into quantifiable signals, ELISA has become an essential tool in clinical diagnostics, biomedical research, and related fields.
Nevertheless, the seemingly straightforward ELISA procedure masks many potential variables. The reliability of qualitative or quantitative results is not automatic; it depends on a thorough understanding of its biochemical principles and strict control of numerous factors throughout the process, from sample handling to data analysis.
This article introduces the basic principles of ELISA and systematically analyzes the key factors that determine experimental success, helping researchers turn this routine method into a reliable foundation for robust and repeatable experiments.
1
What Is ELISA?
ELISA is a commonly used immunological method for detecting and quantifying specific target antigens or antibodies. It depends on the specific binding between antigens and their corresponding antibodies, and achieves qualitative or quantitative analysis through enzyme‑labeled antibodies that trigger chromogenic or chemiluminescent reactions after adding substrates.
ELISA is widely used in many fields, such as basic scientific research, clinical diagnosis (including biomarker detection, disease monitoring, infectious disease testing, allergy screening, and hormone measurement), environmental monitoring, food safety testing, and drug development.
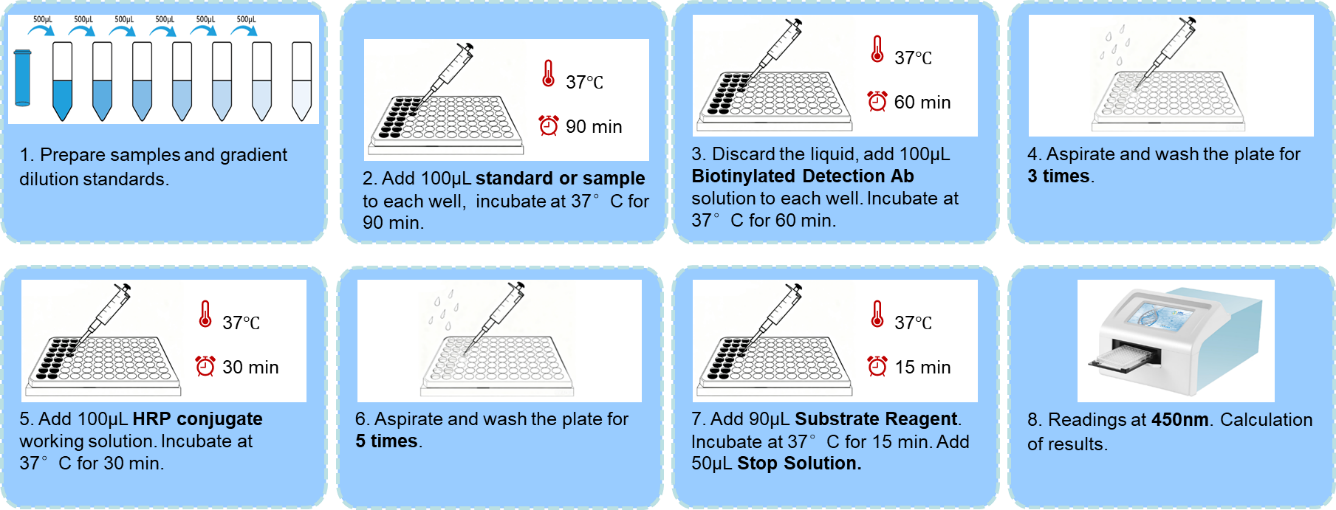
Figure 1. The ELISA technique is used to detect an antigen in a given sample (The antigen is added to the microplate wells and captured by the immobilized antibody. The primary antibody binds specifically to the captured antigen. An enzyme‑labeled secondary antibody is introduced and then catalyzes the color‑producing reaction upon addition of a chromogenic substrate, yielding a color change to quantitatively or qualitatively detect the antigen.)
2
Four Types of ELISA
Based on experimental design and detection goals, ELISA can be divided into four main categories: direct ELISA, indirect ELISA, sandwich ELISA, and competitive ELISA. These types differ in the process and sequence of antigen‑antibody binding. ELISA can also be classified as quantitative or qualitative according to application purposes. The selection of an ELISA format depends on the properties of the analyte and available reagents.
The basic step of ELISA is to immobilize the target antigen or its specific capture antibody onto the surface of microplate wells, allowing direct or indirect detection of the antigen. For high‑sensitivity and high‑specificity measurements, the target antigen can be selectively captured from complex samples using pre‑immobilized antibodies. The captured antigen is then bound by a separate detection antibody, forming an antibody‑antigen‑antibody sandwich structure.
When the target antigen is small or has only one antibody‑binding epitope, a competitive format is used. In this method, labeled antigens compete with unlabeled sample antigens for binding to a limited number of immobilized enzyme‑conjugated primary antibodies. All these ELISA formats can be used for both qualitative and quantitative analysis.
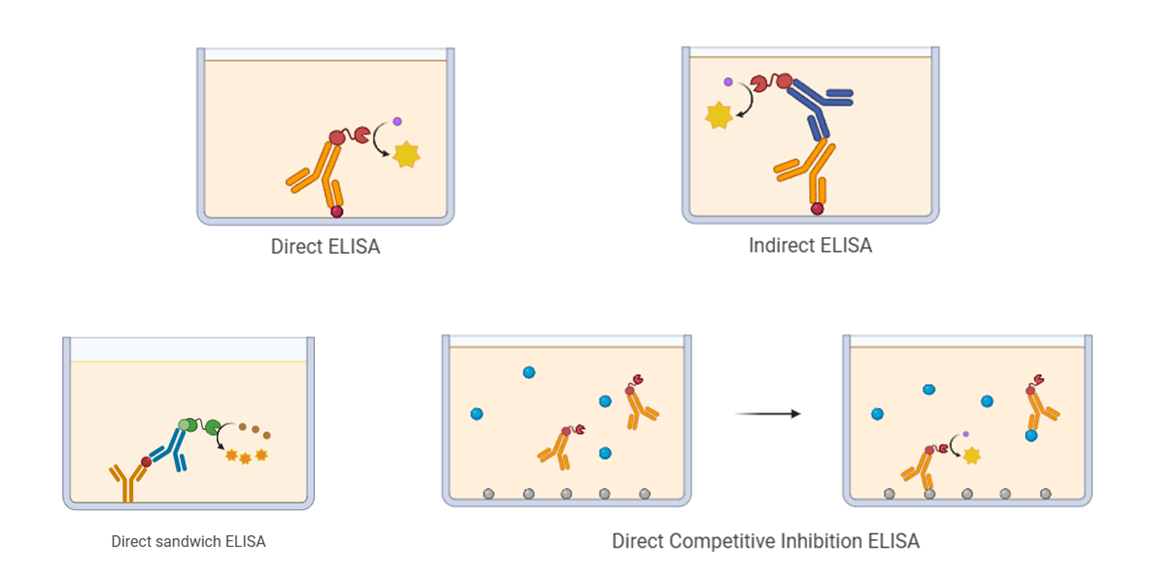
Figure 2. Four Types of ELISA
3
What Factors Could Affect ELISA Results?
ELISA is a multi‑step analytical technique. The accuracy and repeatability of results are affected by a combination of factors across the entire workflow. Inadequate control of these factors can lead to false positives, false negatives, or inaccurate quantification. The main causes of erroneous ELISA results fall into three categories:
• Specimen factor
• Reagent factor
• Operating factor
3.1 Specimen Factors
A wide range of specimens can be used in ELISA, including body fluids (serum, plasma, cerebrospinal fluid), secretions (saliva), and excreta (urine, feces), to detect specific antibodies or antigens. Some samples can be tested directly, while others require pre‑treatment.
Serum is the most frequently used sample in ELISA. False positive and false negative results caused by samples are mainly due to interfering substances, which are divided into endogenous and exogenous types.
| Category | Specific Factors | Description / Impact |
|---|---|---|
| Endogenous Interfering Substances | General Situation | Approximately 40% of human serum contains non‑specific interfering substances that affect results to varying degrees. |
| Rheumatoid Factor (RF) | IgM/IgG‑type RF binds to the Fc region of antibodies in the ELISA system, causing false positives. | |
| Complement | Antibody conformation changes expose the C1q binding site on the Fc region; C1q can bridge the solid‑phase primary antibody and enzyme‑labeled secondary antibody, leading to false positives. | |
| Heterophilic Antibodies | Natural heterophilic antibodies in human serum that bind to rodent immunoglobulins can connect primary and secondary antibodies, causing false positives. | |
| Autoantibodies to Target Antigens | Such as anti‑thyroglobulin and anti‑insulin autoantibodies. They can form complexes with target antigens and interfere with detection. | |
| Iatrogenically Induced Anti‑Mouse Ig Antibodies | Produced by clinical application of mouse monoclonal antibodies or rodent bites. These antibodies may cause false positives. | |
| Cross‑Reactive Substances | Such as digoxin‑like and AFP‑like substances. Effects are minor with polyclonal antibodies but may cause false positives with monoclonal antibodies if cross‑reactive epitopes match. | |
| Other Substances | Excessive serum lipids, bilirubin, hemoglobin, and high blood viscosity all interfere with results. | |
| Exogenous Interfering Factors | Specimen Hemolysis | Releases hemoglobin with peroxidase‑like activity, causing non‑specific color development in HRP‑based ELISA. |
| Bacterial Contamination | Bacteria may contain endogenous HRP, leading to non‑specific coloration. | |
| Improper Specimen Storage | Prolonged refrigeration may cause IgG polymerization or AFP dimerization. Prolonged storage may reduce antigen/antibody activity. Avoid repeated freeze‑thaw cycles. | |
| Incomplete Agglutination | Residual fibrinogen may form clots during testing, potentially causing false positives. | |
| Effects of Tube Additives | Anticoagulants, enzyme inhibitors, and separation gels may interfere with ELISA. |
In summary, when false positives or false negatives occur, sample factors should be investigated first before reagent or operational factors, and corresponding measures should be taken to eliminate interference.
3.2 Reagent Factors (Key Considerations for ELISA Kit Selection)
The quality of commercial ELISA kits varies greatly and can strongly affect detection accuracy, making proper kit selection critical. Key selection criteria include intended application, detection range, applicable sample type, species specificity, and quality control parameters.
| Consideration | Key Points & Explanations |
|---|---|
| Detection Range | The valid interval of the standard curve. Ensure sample concentrations fall within this range. |
| Application Species | ELISA kits for different species are generally not interchangeable unless stated in the instructions. |
| Test Sample Type | Follow kit instructions strictly for sample processing and diluent selection. Do not mix diluents for different samples. |
| Specificity | Determined by the antibody pair. Cross‑reactivity or low affinity reduces sensitivity and specificity. |
| Sensitivity | Reflects the minimum detectable amount of analyte. Select according to expected target abundance. |
| Repeatability | ExKits kits typically achieve intra‑assay CV < 8% and inter‑assay CV < 10%. |
| Literature Citations | ExKits ELISA kits have been cited in more than 27,000 publications. |
| Shelf Life | Kits typically have a shelf life of six months. Check validity when planning experiments. |
3.3 Operational Factors (Key Points for ELISA Experimentation)
ELISA is highly valued in biological research and widely used in clinical testing due to its high sensitivity and specificity. Strictly following the ELISA protocol and paying attention to details is crucial.
| Operational Step | Key Requirements & Precautions |
|---|---|
| Reagent Preparation | Equilibrate all components to room temperature for at least 30 minutes. Mix all reagents thoroughly before use to ensure uniformity. |
| Sample Addition | Process samples in batches, use new pipette tips for each sample to avoid cross‑contamination. Add samples vertically to the bottom of wells. |
| Incubation Control | Strictly follow specified temperature and time. Use a humidified chamber to prevent evaporation and ensure uniform reaction. |
| Washing Step | Follow washing times and soaking duration strictly. Insufficient washing causes high background; excessive washing may reduce signal intensity. |
| Color Development | Protect from light during incubation. Control reaction time accurately to ensure stable and reliable color signals. |
| Result Reading | Read results within 5 minutes after stopping the reaction. Pre‑warm the microplate reader and ensure instrument calibration. |
Conclusion
In summary, ELISA is a powerful and versatile tool in modern bioanalysis, but its successful application requires precision and careful operation. The credibility of final data depends on three interrelated aspects: sample quality, reagent performance, and strict operational control.
By understanding and controlling the key factors discussed, researchers can fully utilize this essential assay. This ensures that results are based on trustworthy, repeatable science, maintaining ELISA’s role as a reliable workhorse technique for years to come.
