The enzyme-linked immunosorbent assay (ELISA), also known as enzyme immunoassay (EIA), represents a cornerstone laboratory methodology that leverages the highly specific interaction between antibodies and antigens, combined with an enzyme-catalyzed colorimetric detection system, to identify and quantify target analytes—including proteins, antibodies, and antigens—across various biological matrices such as serum, plasma, cell culture supernatants, and tissue lysates.
Characterized by exceptional sensitivity (enabling detection of low-abundance targets) and high specificity (minimizing cross-reactivity concerns), ELISA facilitates high-throughput analysis in an economically efficient manner. Its streamlined protocol, modest equipment demands, and excellent reproducibility maintain its broad applicability in clinical diagnostics, biomedical investigation, and bioprocess surveillance.
ELISA permits the detection of a wide array of molecules, encompassing proteins, lipids, carbohydrates, and nucleic acids. It is extensively utilized for measuring serum antibody or antigen concentrations, supporting the diagnosis of infectious diseases stemming from viral, bacterial, and other pathogenic sources. Commercially available ELISA kits enable the detection of numerous human and animal infections, including HIV/AIDS, influenza, cholera, West Nile virus, tuberculosis, and hepatitis B.
Within plant pathology, ELISA functions as an essential diagnostic instrument. Further applications encompass vaccine development, pharmaceutical screening, and at-home pregnancy testing. In the food sector, this assay is applied to detect contaminants such as allergens and toxins, thus safeguarding product safety and integrity.
When you require an ELISA kit for sample analysis, how do you select the appropriate option among the numerous commercially available products? Multiple critical factors warrant careful evaluation when selecting an ELISA kit for your investigative work.
Confirm that the kit accommodates your specific sample type (e.g., serum, plasma, urine, tissue homogenates, cell culture supernatant). Distinct sample matrices may necessitate particular handling procedures.
The majority of commercial ELISA kits are validated using serum/plasma and culture supernatants. Careful review of product documentation is essential to ensure compatibility with your samples. For instance, plasma collection methodology (heparin versus EDTA) can influence kit selection.
Additionally, factors including hemolysis and lipid content may compromise assay performance. These considerations should be evaluated prior to kit selection. Acquiring a trial-size ELISA kit to conduct preliminary testing with your samples is advisable to confirm kit suitability and sensitivity for your specific matrix.
Determine the total number of samples requiring analysis and select kit quantities accordingly. ELISA kits typically require sample volumes ranging from 100μL down to 10μL. When sample volume is severely limited or samples are particularly valuable, prioritize ELISA kits with minimal sample volume requirements.
While most providers supply 96-test ELISA kits, certain manufacturers, such as ExKits, also offer 24-test trial ELISA kits.
Recovery and linearity assessments serve to evaluate ELISA kit performance. Recovery analysis determines whether analyte detection is influenced by variations in sample matrices. Superior recovery values indicate better performance. Linearity of dilution establishes the degree to which analyte dose-response remains linear within a specific diluent. Ideally, sample concentrations should remain consistent across all dilutions.
Most providers include recovery and linearity data within product specifications. Additional critical parameters, including sensitivity and dynamic range, are also typically provided. Performance parameters may vary among manufacturers. Careful comparison of these metrics, particularly recovery and linearity data, will inform appropriate kit selection.
Appropriate ELISA format selection is crucial for experimental success, as each configuration is optimized for specific detection objectives. The fundamental distinctions among formats center on antigen-antibody interaction mechanisms and signal generation strategies. Your selection should be primarily informed by target analyte molecular dimensions and your sensitivity and specificity requirements.
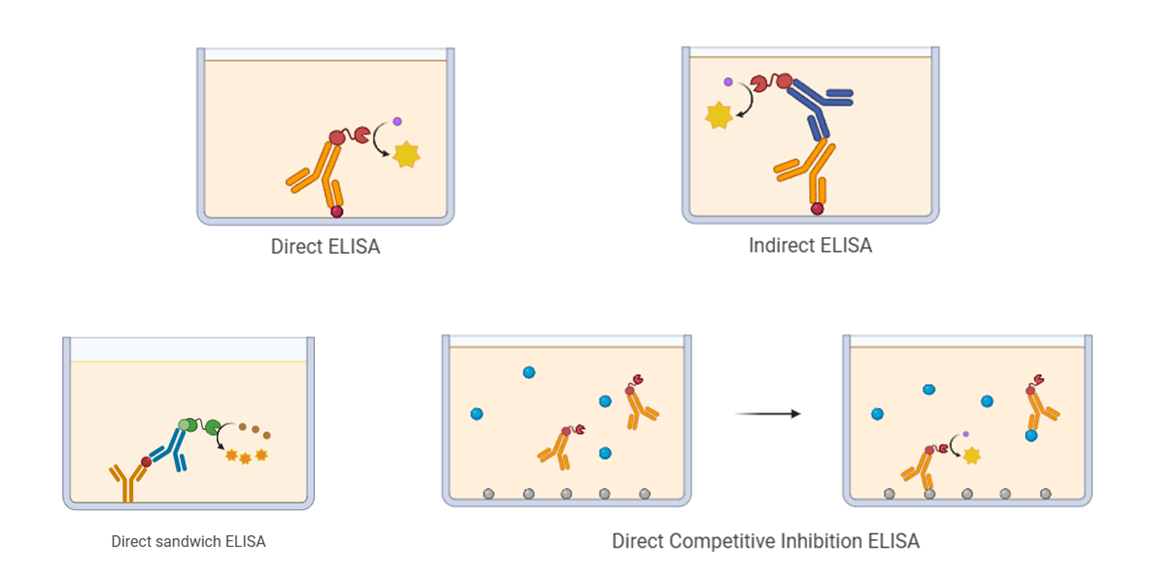
| ELISA Format | Primary Applications | Defining Feature | Sensitivity Level | Signal Correlation | Benefits | Limitations |
|---|---|---|---|---|---|---|
| Direct ELISA | Large, abundant antigens | Single enzyme-conjugated primary antibody | Low | Positive | - Rapid and straightforward - Reduced cross-reactivity risk | - Reduced sensitivity - Limited flexibility |
| Indirect ELISA | Antibody detection (e.g., serological applications) | Unlabeled primary antibody with enzyme-conjugated secondary antibody | High | Positive | - Enhanced sensitivity - Economical and adaptable | - Elevated background potential - Extended procedure duration |
| Sandwich ELISA | Large antigens with multiple epitopes (e.g., cytokines) | Dual antibody system targeting distinct sites | Highest | Positive | - Superior specificity and sensitivity - Compatible with complex matrices | - Antigen structural requirements - Matched antibody pair necessity |
| Competitive ELISA | Small molecules/haptens (e.g., hormones, pharmaceuticals) | Sample analyte competes with reference standard for antibody binding | Moderate | Inverse | - Optimal for small molecules - Tolerant of impure samples | - Inverse signal interpretation - Moderate sensitivity and specificity |
| QC Parameter | Definition | Optimal Acceptance Criteria |
|---|---|---|
| Standard Curve Detection Range | The concentration span across which the assay accurately quantifies the analyte | Standard curve correlation coefficient (R²) should exceed 0.99 |
| Limit of Detection (LOD) / Sensitivity | The minimum analyte concentration reliably distinguishable from blank samples | Lower values indicate superior sensitivity (e.g., pg/mL or fg/mL levels) |
| Linearity | Assay capacity to produce results directly proportional to analyte concentration in diluted samples | Dilution linearity should fall within 80-120% |
| Coefficient of Variation (CV) / Precision | Repeatability measure for identical samples. Intra-assay CV assesses within-plate variation; Inter-assay CV evaluates between-plate/run variation | Intra-assay CV: < 8-10%; Inter-assay CV: < 10-15% |
| Recovery (Spike and Recovery) | Accuracy of measuring known analyte quantities (spikes) added to samples, expressed as percentage of expected value | Typically 80-120%; indicates minimal matrix interference |
| Stability | Kit capacity to maintain performance characteristics throughout specified storage conditions | Evaluated through accelerated stability testing (e.g., 37°C for 7 days); performance deviation should remain < 15% |
| Specificity | Kit capacity to exclusively detect target analyte without cross-reactivity with related proteins or matrix interference | High specificity confirmed by minimal or absent cross-reactivity with homologous proteins or common serum components |
| Natural Sample Validation | Demonstration of accurate kit performance with authentic biological samples, not merely buffer solutions | Accurate data generation across various sample types (serum, plasma, etc.) while avoiding complications from contaminated or hemolyzed specimens |
| Evaluation Criterion | Assessment Parameters | Significance |
|---|---|---|
| Manufacturer Profile | Corporate history, operational scale, certifications (e.g., ISO 9001), and core competencies | Establishes fundamental reliability, production capacity, and compliance with international quality standards |
| Technical Support Services | Availability of pre-purchase consultation, experimental customization options, and responsive post-purchase assistance | Ensures appropriate kit selection, optimal application, and timely problem resolution, conserving valuable research resources |
| Reputation and User Feedback | Independent evaluations, publication citations, and peer community feedback | Provides empirical evidence of performance, reliability, and manufacturer standing within the scientific community |
| Literature Citations | Frequency of kit utilization and citation in peer-reviewed scientific literature | Extensive citation patterns indicate validation, reliability, and broad expert acceptance. |
| Cost-effectiveness | Total cost relative to performance, encompassing per-test pricing, repeat experiment rates, and potential costs of failed experiments | Balances budgetary constraints against data reliability requirements, emphasizing long-term value over initial expenditure |
